COVISTIX™ and COVIMARK™* (Antigen Test for the Detection of SARS-CoV-2 Virus in Nasal Swab)
SIMPLE:
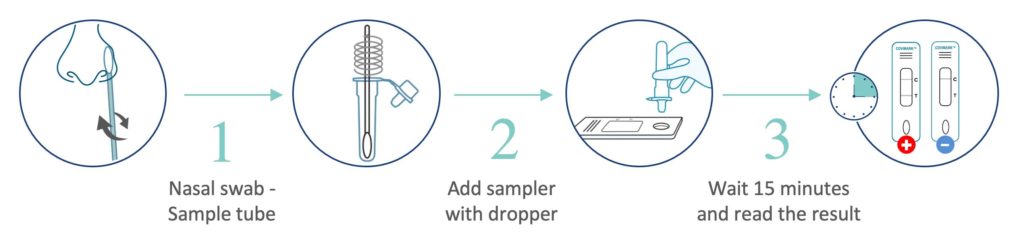
3-step test procedure
RAPID:
Produces Results in 15 minutes or less
CONVENIENT:
Nasal swab with simple 3 step instructions and visible read
ACCURATE:
Highly sensitive platinum colloid-based lateral flow N-Antigen immunoassay to detect SARS-CoV-2 virus (Detects Omicron and Omicron Subvariants)
* Trademarked as COVISTIX™ in Mexico and Brazil. COVIMARK™ in all other Geographies

