RTX
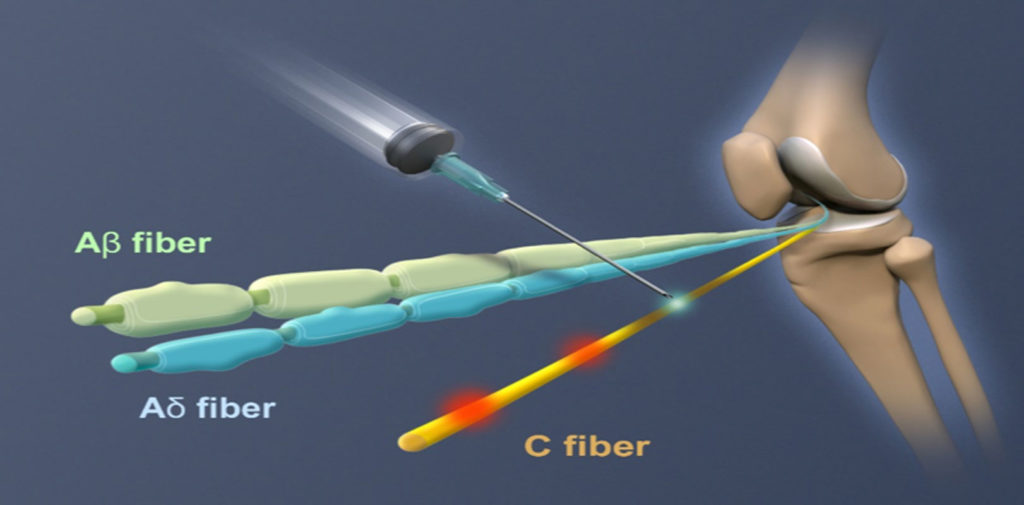
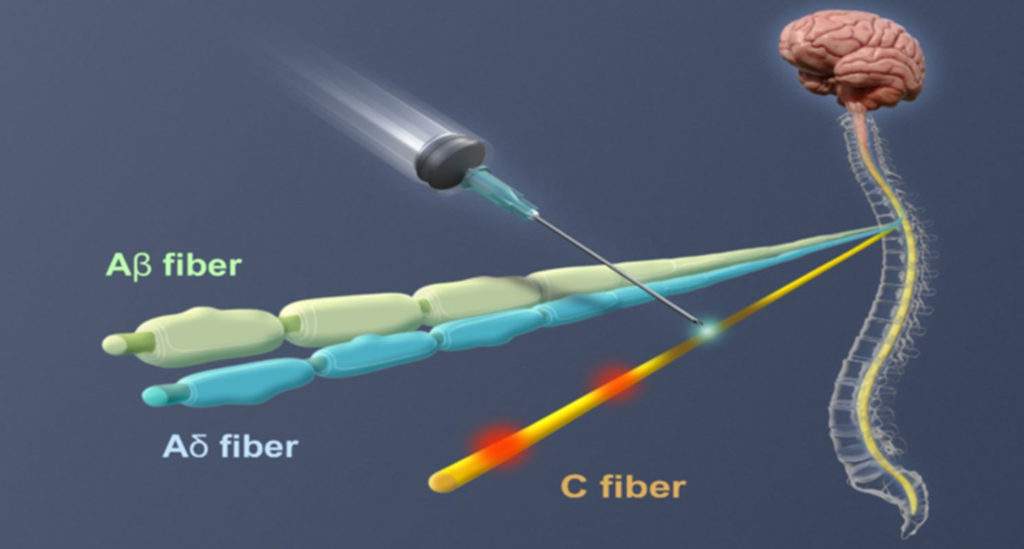
RTX (resiniferatoxin) is a unique neural intervention molecule that is highly selective and may be applied peripherally (e.g., nerve block, intra-articular) or centrally (e.g., epidural), to control chronic pain across multiple conditions including arthritis and cancer.
RTX has the potential to be a first-in-class drug addressing currently intractable pain in a novel and unique way, by targeting the nerves responsible for the chronic debilitating pain signal transmission.
RTX strongly binds to TRPV1 receptors and forces open calcium channels located in the end-terminal of the nerve or the soma of the neuron (depending upon the route of administration). This in turn generates a slow and sustained cation influx that rapidly leads to the deletion of TRPV1-positive cells.
RTX directly interacts with afferent nerve cells without affecting sensations such as touch, pressure, acute prickling pain, vibration sense or muscle coordination function.
Administration at the peripheral nerve ending results in a sustained temporal effect to treat pain associated with arthritis of the knee.
RTX can potentially help patients with terminal cancer pain, after a single epidural injection, by permanently blocking the pain signal transmission from the tumor tissue to the dorsal root ganglion (DRG) in the spinal cord, without the undesirable side effects associated with high and repeated doses of opioids. If opioids remain part of the therapeutic arsenal for these patients, RTX has the potential to significantly reduce the amount and frequency of opioid use.
RTX has been granted Orphan Drug Status by the US Food and Drug Administration for the treatment of end-stage diseases, including intractable cancer pain.
Sorrento has successfully completed a positive Phase Ib clinical proof of concept trial with the National Institutes of Health under a Cooperative Research and Development Agreement (CRADA) which showed improved pain and reduced opioid consumption after intrathecal administration (directly into the spinal cord space).
The company has initiated pivotal studies and is aiming for an NDA filing in 2024.

